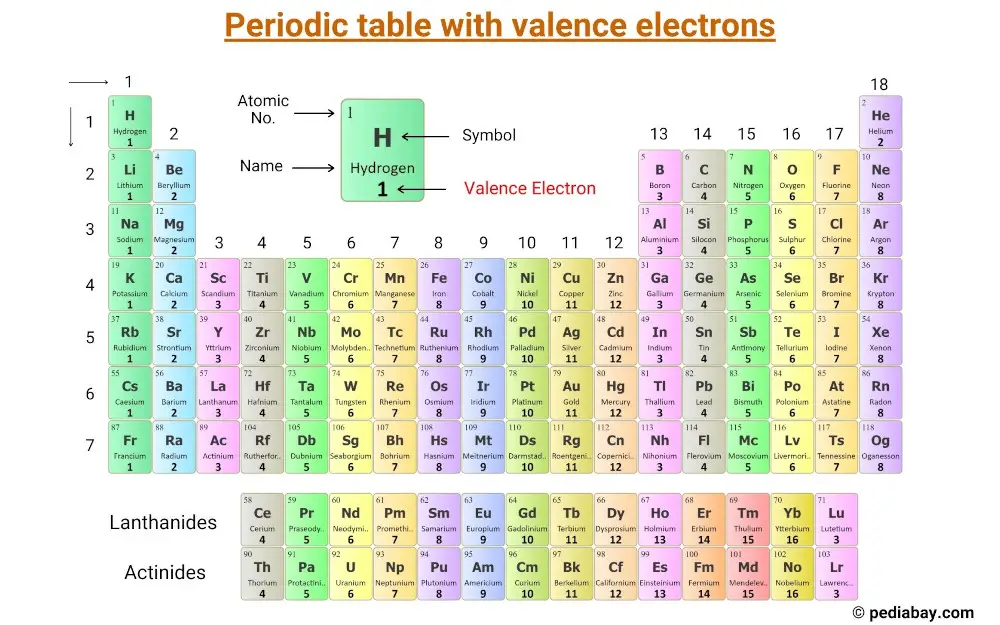 
(Atoms have protons and neutrons in their nucleus, and surrounding that, they have their electrons arranged in orbitals, where an atomic orbital is a math term that describes the location of an electron as well as its wave-like behavior.)įor instance, period 1 includes elements that have one atomic orbital where electrons spin period 2 has two atomic orbitals, period 3 has three and so on up to period 7. So in general, I would say the number of valence electrons for transition metals (and lanthanide and actinides) vary in an unpredictable way, but the valence orbitals could sometimes be predicted with enough chemical intuition.ĭISCLAIMER: It is, however, in general difficult for the lanthanides and actinides to predict which orbitals are valence.įor example, the actinides have #5f# and #6d# orbitals very close in energy to their #7s# orbital, so we may GUESS and include the #7s#, #6d#, AND the #5f# in the valence space (even if the #6d# orbitals are empty) when performing atomic energy computations.The horizontal rows on the periodic table are called periods, where each period number indicates the number of orbitals for the elements in that row, according to Los Alamos National Laboratory. On the other hand, we could easily say that the valence ORBITALS of the first-row transition metals are the #4s# and #3d# orbitals. 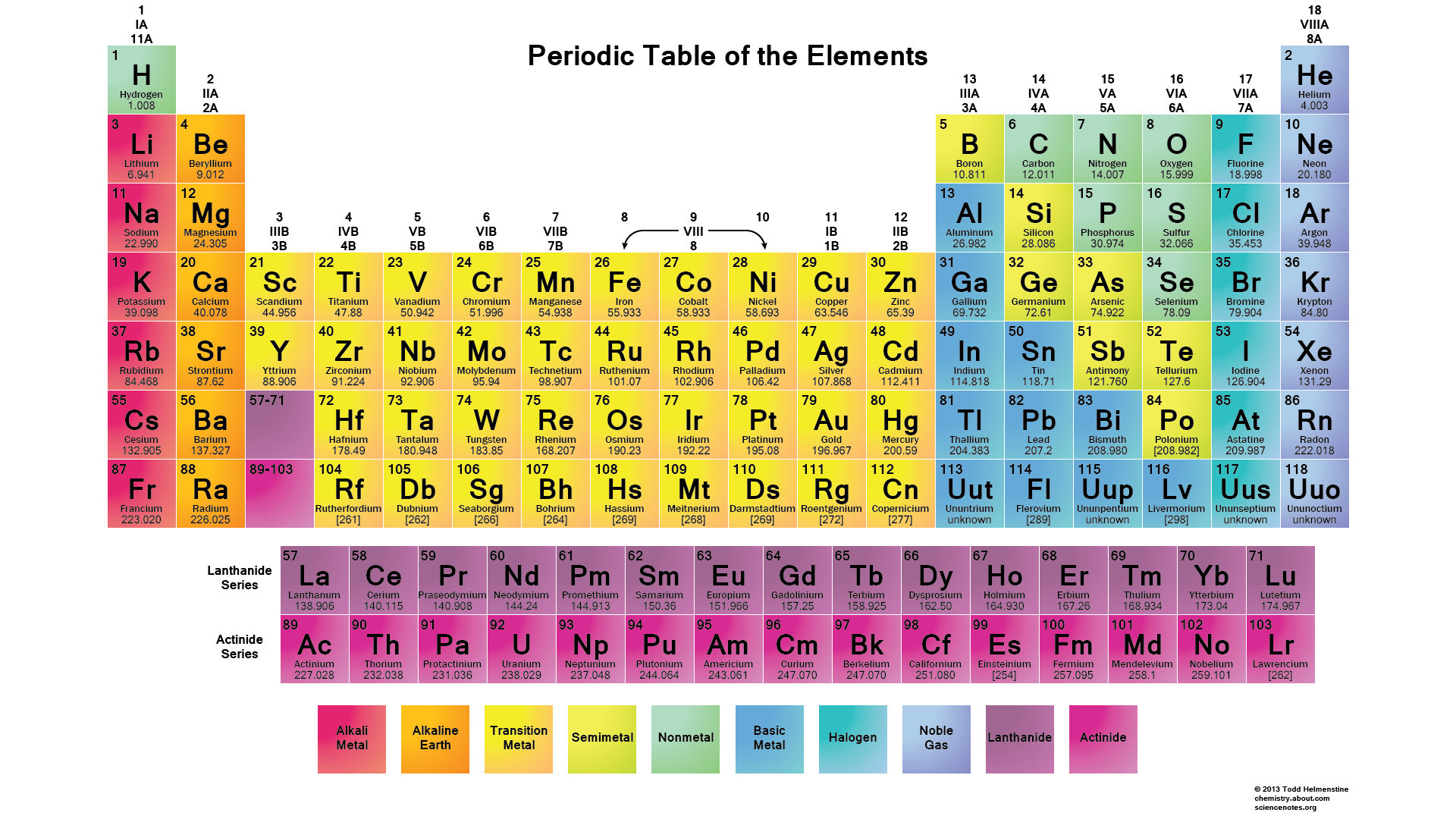
#"CuCl"_2#), and so it is capable of taking from its #bb(3d)# electrons as well for its valence electron(s). But a #+2# oxidation state is also known (e.g. #"CuCl"#), so it makes sense that it uses its one #4s# electron most often as its valence electron(s). Copper tends to have a #+1# oxidation state (e.g.Chromium could have up to six valence electrons, which would include its #3d# electrons, since it can accomplish a #+6# oxidation state (i.e.A #+3# oxidation state would have required transferring three valence electrons if it were to form a pure cation. Scandium makes sense to have up to three valence electrons, since a #"Sc"^(+3)# oxidation state exists (e.g.In general, the first-row transition metals have a set of valence orbitals that include their #4s# and #3d#'s, but the number of valence electrons will vary. It is easier and more practical to describe which orbitals are valence orbitals when it comes to transition metals (although it gets difficult with lanthanides and actinides). For transition metals, the word "important" will vary depending on the context. Valence electrons are those that are important in chemical bonding. Having a filled 3d and a half fille 4s subshell is more stable than # 4s^2 3d^9#. Copper has one valence electron (the 4s electron) because it has electron configuration of # 4s^1 3d^10#. Chromium has an electron configuration of # 4s^1 3d^5# because having a half filled 3d subshell is more stable, so it has one valence electron. The two 4s electrons are in the highest principal quantum number, n = 4, so they are the valence electrons.Ĭopper and chromium have one valence electron (they are exceptions), because they have one 4s electron. Reason: The electron configuration of Fe is #1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^5#. How many valence electrons are there in Fe? Most transition metals have an electron configuration that is #ns^2 (n-1)d#, so those #ns^2# electrons are the valence electrons.įor example. Valence electrons are the sum total of all the electrons in the highest energy level (principal quantum number n). Most transition metals have 2 valence electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
